The renin-angiotensin-aldosterone system (RAAS) is a vital regulatory system involved in several physiological processes, including blood pressure regulation and fluid balance. A key player in this intricate system is angiotensin-converting enzyme 2 (ACE2), an enzyme that counteracts the effects of the classical RAAS pathway. Recent research has shed light on the potential of ACE2 as a therapeutic target, leading to the development of the ACE2 Library. In this article, we explore the role of ACE2 in RAAS regulation and the promise of the ACE2 Library in unlocking new avenues for drug discovery and therapy.
Understanding RAAS Regulation:
The RAAS pathway involves a cascade of biochemical reactions that regulate blood pressure, fluid balance, and electrolyte homeostasis. Central to this system is angiotensin II (Ang II), a hormone that constricts blood vessels and promotes sodium and water retention. Ang II is generated by the action of angiotensin-converting enzyme (ACE) on angiotensin I. However, ACE2 offers an alternative pathway by enzymatically degrading Ang II into Ang-(1-7), which opposes the effects of Ang II.
Role of ACE2:
ACE2 plays a pivotal role in counterbalancing the harmful effects of Ang II in the RAAS pathway. By degrading Ang II and producing Ang-(1-7), ACE2 exerts protective effects on the cardiovascular system. Ang-(1-7) promotes vasodilation, reduces inflammation and fibrosis, and improves endothelial function. ACE2 also has a positive impact on other organs, as it is expressed in various tissues, including the lungs, kidneys, heart, and gastrointestinal tract.
The Promise of the ACE2 Library:
Recognizing the therapeutic potential of modulating ACE2 activity, researchers have developed the ACE2 Library—a collection of small molecule compounds designed to target ACE2, either by enhancing its activity or inhibiting its degradation. The library offers a valuable resource for drug discovery and the development of novel therapeutics for cardiovascular diseases, respiratory illnesses, and other conditions impacted by disruptions in the RAAS system.
Implications for Cardiovascular Diseases:
Cardiovascular diseases, such as hypertension, myocardial infarction, and heart failure, are often associated with dysregulated RAAS activity. The ACE2 Library presents an opportunity to regulate the RAAS system and potentially alleviate the effects of these diseases. Manipulating ACE2 activity could provide a novel approach to improving cardiovascular health by reducing Ang II levels, promoting vasodilation, and modulating inflammation and fibrosis.
Respiratory Applications and Beyond:
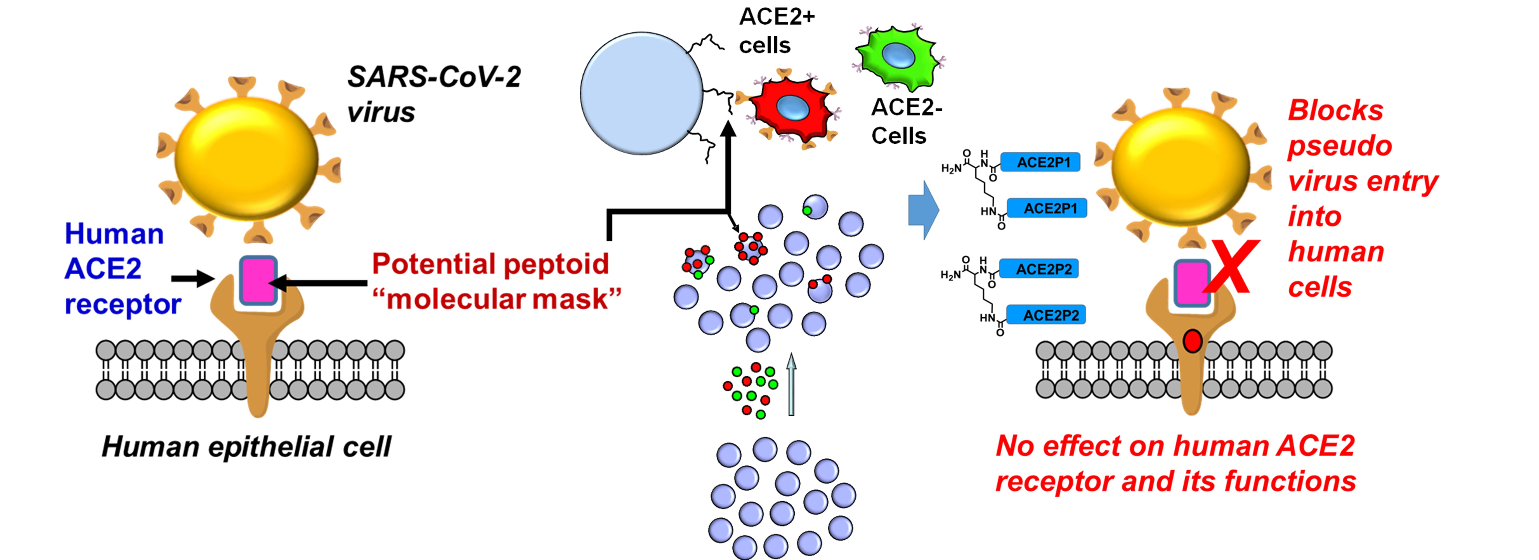
Beyond cardiovascular diseases, the ACE2 Library holds promise in respiratory conditions, particularly in light of the COVID-19 pandemic. ACE2 serves as the entry receptor for the SARS-CoV-2 virus, which causes COVID-19, leading to concerns about potential ACE2-targeted therapies. However, the ACE2 Library could aid in identifying compounds that enhance ACE2 activity while minimizing susceptibility to viral entry, providing potential therapeutic avenues for respiratory diseases.
Future Directions and Challenges:
While the ACE2 Library offers exciting possibilities, challenges remain. Developing compounds that selectively target ACE2 without interfering with its physiological functions or promoting off-target effects is a crucial consideration. Rigorous preclinical and clinical testing will be necessary to assess the safety and efficacy of potential ACE2 modulators. Moreover, understanding the complexity and dynamic regulation of the RAAS system will be essential for successful drug development.
Conclusion:
The ACE2 Library and its potential as a therapeutic resource mark an important advancement in RAAS regulation. By targeting ACE2, researchers aim to rebalance the RAAS system and combat cardiovascular diseases, respiratory conditions, and other associated disorders. As research in this field continues to progress, the ACE2 Library offers a valuable platform for drug discovery, bringing us closer to unlocking the full potential of ACE2 and revolutionizing the treatment of RAAS-related conditions.